DE Outbreak of Lung Injury Associated with E-Cigarette Use, or Vaping
The Delaware Division of Public Health continues to participate in a multi-state investigation into a national outbreak of severe pulmonary disease. As of Jan. 14, 2020, the CDC reports 2,668 hospitalized cases or deaths. The investigation includes such vaping-associated products as devices, liquids, refill pods, and cartridges. A total of 60 deaths have been confirmed in 27 states and the District of Columbia. Additional details on the national investigation can be found on the CDC’s website.
This webpage will provide ongoing updates regarding the outbreak, Delaware’s response to it, and our recommendations.
Delaware has 20 cases of e–cigarette, or vaping, product use–associated lung injury (EVALI) that meet the CDC’s case definition of either probable or confirmed. Sadly, in one of the cases associated with this national outbreak, an individual died. Some information about Delaware’s 20 cases:
- 13 are male, 7 are female
- The age range of patients is between 15 to 65 years old. *Note: This doesn’t mean the youngest is 15 and the oldest is 65, but that the patient’s ages fall within this range.
- The average age range of the patients is 28 years old
- Twelve of the individuals are from New Castle County, three are from Kent and five are from Sussex
- Five of the patients report using e-cigarette products containing THC alone; eight report using THC in combination with nicotine e-cigarettes (One of the reported uses of THC products included vaping medical marijuana.) Seven of the 20 persons reported using e-cigarette products containing nicotine only.
The Latest
- CDC, FDA, and state health authorities have made progress in identifying substances of concern in EVALI, and in characterizing the outbreak.
- Emergency department (ED) visits related to e–cigarette, or vaping, products continue to decline, after sharply increasing in August 2019 and peaking in September.
- National and state data from patient reports and product sample testing suggest tetrahydrocannabinol (THC)–containing
e–cigarette, or vaping, products, particularly from informal sources like friends, family, or in–person or online
dealers, are linked to most EVALI cases and play a major role in the outbreak.
- 82% of hospitalized patients with data on substance use reported using THC–containing products; 33% reported exclusive use of THC–containing products (as of January 14, 2020).
- 50% of EVALI patients who reported using THC–containing products provided data on product source (as of January 7, 2020)
- 16% reported acquiring products only from commercial sources (recreational and/or medical dispensaries, vape or smoke shops, stores, and pop-up shops).
- 78% reported acquiring products only from informal sources (family/friends, dealers, online, or other sources).
- 6% reported acquiring products from both commercial and informal sources.
- CDC and FDA recommend that people not use THC–containing e–cigarette, or vaping, products, particularly from informal sources like friends, family, or in-person or online dealers.
- In an Illinois study, 14% of 121 interviewed EVALI patients reported using only nicotine–containing e–cigarette, or vaping, products. This is consistent with national findings.
- Nine of the interviewed EVALI patients had no indication of any THC use.
- Some EVALI patients report the use of only nicotine-containing e–cigarette, or vaping, products. The contributing cause or causes of EVALI for individuals reporting use of only nicotine-containing products warrants further study.
- Vitamin E acetate is strongly linked to the EVALI outbreak. Vitamin E acetate has been found in product samples tested by FDA and state laboratories and in patient lung fluid samples tested by CDC from geographically diverse states. Vitamin E acetate has not been found in the lung fluid of people that do not have EVALI.
- However, evidence is not sufficient to rule out the contribution of other chemicals of concern, including chemicals in either THC or non–THC products, in some of the reported EVALI cases.
CDC will continue to update guidance as it learns more about EVALI.
What is Vaping-Associated Lung Illness?
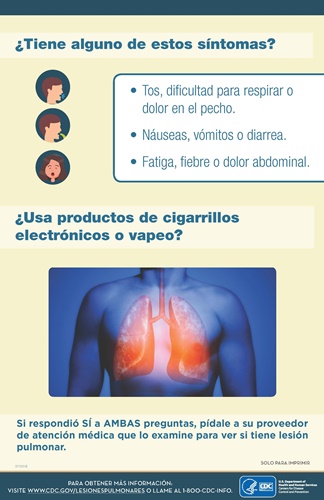
As seen in this outbreak, vaping can be deadly. VAPI symptoms typically develop over a period of days, but sometimes can manifest over several weeks. Symptoms include:
- Rapid heart beat (regular or irregular)
- Shortness of breath
- Nonproductive cough
- Pleuritic chest pain
- Fever
- Chills
- Fatigue
- Gastrointestinal distress: nausea, vomiting, abdominal pain, diarrhea
- Hypoxemia (abnormally low blood-oxygen level)
- Acute or subacute respiratory failure
As in the national cases, no single product or device in Delaware has been identified in all cases, but we do know that use of vaping or e-cigarette products containing either THC alone or in combination with nicotine products is reported in most cases.
Although vaping proponents advocate that these products have helped them quit using combustible cigarettes, these products are not recognized by federal health agencies as approved smoking cessation devices. Many people falsely believe that vaping is healthier than smoking, when in fact neither is considered safe.
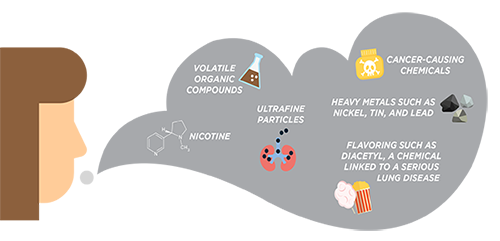
According to the Centers for Disease Control and Prevention (CDC), although e-cigarette aerosol generally contains fewer toxic chemicals than regular cigarettes, e-cigarette aerosol can contain harmful and potentially harmful substances, like nicotine, heavy metals like lead, volatile organic compounds, and cancer-causing agents. Additionally, some e-cigarette products contain as much nicotine as 20 cigarettes.
What is DPH Doing?
On August 20, DPH began sharing the CDC’s information on this evolving outbreak to health care providers through Health Advisory Notices (HANs). Recommendations to providers included asking them to report suspected cases to DPH for investigation. A small number of reports began coming in the next day. DPH appointed a lead epidemiologist to follow up on suspect cases, and additional epidemiologists are searching medical databases for possible cases.
On August 28, DPH started reporting cases that met the CDC case definition of “probable” to the CDC. A second HAN with updated CDC information was shared with the medical community on Aug. 30. DPH issued a press release on Sept. 9, announcing the investigation of three possible cases, and issued a follow-up release on Sept. 25. On Oct. 3, DPH held a media call to provide an update and announce Delaware’s first death associated with this outbreak.
Delaware has submitted samples of vaping devices to the FDA for testing. DPH does not have any results at this time from FDA testing.
We also have asked the state’s medical marijuana compassion centers to post signage advising patients to consider if vaping is the best option for their medical use, and to have a thoughtful conversation with their health care provider or compassion center staff about possibly safer alternatives to vaping.

Recommendations for the Public
- DPH strongly encourages people not to use e-cigarette products, particularly those containing THC, whether the THC products are legal or illegal. The use of regulated THC products has been reported in cases of severe lung injury in other states.
- People who continue to use e-cigarette products should monitor themselves for symptoms including: coughing, shortness of breath, chest pain, nausea, vomiting, abdominal pain, and fever – and promptly seek medical attention for any health concerns.
- People with underlying chronic respiratory conditions are particularly susceptible to increased effects or impacts of using e-cigarette products, and should stop using them immediately, or not start using e-cigarette products in the first place.

- Although there is risk with any vaping product, people should not buy e-cigarette products off the street and should not modify those products or add any substances that are not intended for use by the manufacturer.
- E-cigarette products should never be used by youth, young adults, pregnant women, or adults who do not currently use tobacco products.
- Those individuals who switched from combustible cigarettes to e-cigarettes and discontinue vaping, should not return to smoking combustible cigarettes. The Division of Public Health recognizes that there are people who have chosen to use e-cigarette products as a step-down from using combustible tobacco products, but the FDA does not recognize the use of e-cigarette products as an approved smoking cessation product.
- Instead if you are an adult who needs help quitting tobacco products, including e-cigarettes, call the Delaware Quitline at 1-866-409-1858. Youth who smoke or vape can contact the American Lung Association’s N-O-T (Not on Tobacco) Cessation Program for teens at 1-800-LUNGUSA. The Truth Initiative also operates a text cessation program for youth. To participate, text “DITCHJUUL” to 887-09.
- If you are using vape-/e-cigarette products with THC for medical purposes, contact your health care provider and have a conversation about potentially safer alternatives for you.
- If you are vaping illegal THC products and need help stopping the use of marijuana, contact the Division of Substance Abuse and Mental Health’s Crisis Line. In New Castle County, the number is 1-800-652-2929 and in Kent and Sussex counties, it is 1-800-345-6785.
For Health Care Providers
The CDC issued updated interim guidance in late November to guide health care providers as rates of influenza in the community increase. The CDC advises providers evaluating patients with respiratory illnesses to ask them about e-cigarette, or vaping, product use; evaluate whether patients require hospital admission; and consider empiric use of antimicrobials, including antivirals, as well as possible corticosteroids. Vaping associated lung injury (VAPI) is a diagnosis of exclusion; rapid recognition of VAPI (also known as EVALI) patients by health care providers is critical to reducing severe outcomes. This information was published as a CDC Morbidity and Mortality Weekly Report (MMWR), which includes an algorithm for management of patients with respiratory, gastrointestinal, or constitutional symptoms and e-cigarette, or vaping, product use.
DPH and the CDC recommend health care providers:
- Report cases of lung injury of unclear etiology and a history of e-cigarette or vaping product use within the past 90 days to DPH at 1-888-295-5156. Reporting of lung injury cases may help the CDC and state health departments determine the cause of these lung injuries.
- Screen all patients for e-cigarette or vaping product use, including nicotine and THC containing products.
View additional recommendations here: https://www.cdc.gov/tobacco/basic_information/e-cigarettes/severe-lung-disease/healthcare-providers/index.html

For the coming flu season, CDC recommends that clinicians strongly consider respiratory infections such as influenza, as well as lung injury associated with e-cigarette use, or vaping, in all patients presenting with respiratory symptoms and a history of e-cigarette use, or vaping.
Clinicians are further advised that it may not be possible to distinguish definitively between respiratory infection and lung injury associated with use of e-cigarettes, or vaping, using available testing. Therefore, therapy directed at both may be necessary. Decisions on the initiation or discontinuation of treatment should be based on specific clinical features and, when appropriate, in consultation with specialists.
DPH also asks that providers download this poster (Spanish available too) and place it in their waiting and patient rooms as space allows:
Resources:
Additionally, providers can remain informed about the latest updates via the CDC’s lung injury webpage: https://www.cdc.gov/lunginjury.
The risks of e-cigarette use: https://dhss.delaware.gov/dph/
![]() Please note: Some of the files available on this page are in Adobe PDF format which requires Adobe Acrobat Reader. A free copy of Adobe Acrobat Reader can be downloaded directly from Adobe . If you are using an assistive technology unable to read Adobe PDF, please either view the corresponding text only version (if available) or visit Adobe’s Accessibility Tools page.
Please note: Some of the files available on this page are in Adobe PDF format which requires Adobe Acrobat Reader. A free copy of Adobe Acrobat Reader can be downloaded directly from Adobe . If you are using an assistive technology unable to read Adobe PDF, please either view the corresponding text only version (if available) or visit Adobe’s Accessibility Tools page.